Houston antibody trials underway in midst of Trump's ailment

Just as President Trump was receiving a dose of an experimental antibody drug from Regeneron Pharmaceuticals, UTHealth in Houston was signing up its first participant for a trial of a similar antibody treatment from drug maker Eli Lilly.
"There's been a history of using antibody's to treat infections," said Dr. Netanya Utay, Associate Professor of Internal Medicine at UTHealth. "The results that are out so far definitely look promising, but they're from small trials. We still need to enroll a lot more patients in these studies to really determine how effective they are for decreasing time to symptom resolution and decreasing hospitalizations."
She says several drug makers are developing laboratory made antibodies to help boost the body's response to COVID-19 infection.
The goal is to minimize symptoms and length of sickness.
UTHealth is conducting several trial studies for antibody therapies from different drug makers.
One trial is for family members of COVID-19 positive patients.
"So it's for people who don't have symptoms and who have tested negative for COVID-19 but are living with someone who does have COVID-19," said Dr. Utay.
This treatment is from Regeneron, the same company behind the experimental drug given to Trump.
SEE MORE: Possible COVID-19 preventative is in clinical trials

"He's in a race where his immune system is racing against the virus and if the virus wins you can have dire consequences obviously. And what our antibodies do is we make it a fairer fight," said Dr. Len Schleifer, Co-Founder and CEO of Regeneron. "It's a cocktail of two monoclonal antibodies that work together to really glum on to that virus and effectually make it impossible for the virus to get in your cells."
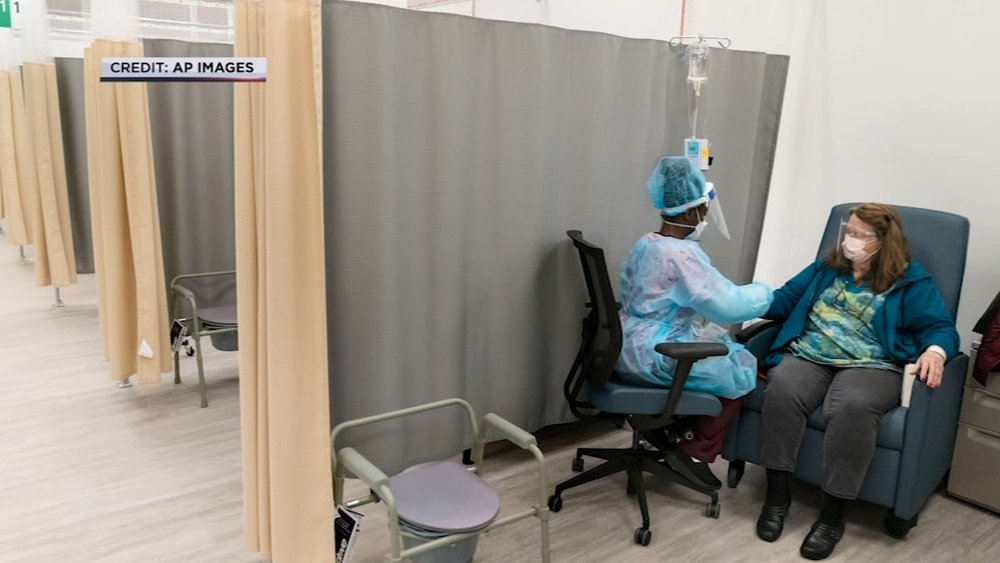
UTHealth is also looking for COVID-19 positive patients, who have mild symptoms for an antibody treatment from Eli Lilly.
"The infusion takes about an hour and then they're monitored for about two hours after that to make sure they don't have any adverse reactions," said Dr. Utay.
Both studies are being run out of LBJ Hospital.
If you're interested in these trials, you can call 409-234-1398.